No part of the proposed FDA regulations regarding cigars has been more talked about than a proposed $10 price point for exemption. Despite all this talk—and outrage—the reality is, that one sentence is almost certain to change by the time a law becomes effect, likely due to something other than the $10 itself.
Click here for a more complete breakdown of the proposed regulations.
For those not caught up with the discussion over the U.S. Food and Drug Administration’s deeming document, the basics are as follows.
The FDA is finally ready to extends its authority over tobacco products it does not currently regulate, mainly cigars and e-cigarettes. However, it is not sure whether to regulate all types of cigars and as such has sought public comment on which cigars to regulate.
Under option one, all cigars would be subject to new regulation including new compliance laws, disclosure of ingredients and manufacturing process, new warning labels, a ban on samples and other restrictions.
Under option two, the FDA would exempt “premium cigars.” Part of the proposed exemption includes a discussion of what should be exempted, or more specifically, what’s a “premium cigar.”
There are eight proposed standards that a product must meet in order to be a premium cigar, one of which—point six—deals with price.
The exact texts reads:
6. Has a retail price (after any discounts or coupons) of no less than $10 per cigar (adjusted, as necessary, every 2 years, effective July 1st, to account for any increases in the price of tobacco products since the last price adjustment);
The great thing is, the FDA is asking for public comment specifically on both issues: a. whether it should exempt premium cigars, b. what is a “premium cigar.” The reality is, if it goes down this route, it’s almost impossible to believe that the text of standard six isn’t going to change.
1. THE REGISTER PRICE WILL NOT BE THE STANDARD USED
There’s a reason why when states tax cigars, they tax based off of the wholesale price. There’s an established inventory and a much more consistent paper trail than what goes on at the register.
Furthermore, it would make the regulation entirely subjective.
One retailer could sell a cigar at $9.50, while a retailer down the road could sell it at $10—even though the second retailer was selling a cigar that was presumably in the clear as far as price goes.
Yet how the rules are written, the first retailer would presumably ruin it for everyone—that $9.50 cigar would fall below the final price requirement and as such subject to FDA regulation if the exemption passes. Would the FDA then go back and add that cigar to the list of products to regulate? It’s hard to imagine it doing so one cigar at a time.
Think the “down the road” example is to small? Just imagine what state and local taxes will do to enforcement. Cigars in states like Florida, New Hampshire and Pennsylvania—where premium cigars are not subject to a state tobacco tax—are cheaper, a lot cheaper. In some cases a cigar that retails for $6 in those three states is in fact a $10 cigar in Utah simply because of an 83 percent state tobacco tax. Does that cigar become exempt because it’s $10 in Utah? Or does the FDA regulate it because it’s not $10 in most humidors in the country.
The amount of control the FDA has once the cigars leave a distributor is greatly reduced. In order for a law to be enforced, the FDA is likely going to have to monitor things at the manufacturer and distributor level, not at the register.
That leaves two options for a price standard. One, the way just about every state taxes cigars, is based off of the wholesale or “manufacturer’s price”—i.e. the price paid by distributors or retailers to the manufacturers. The other option is to base the price requirement off of the manufacturer’s suggested retail price (MSRP).
2. AND IT WILL NOT BE MSRP
In addition to the better paper trail created by the wholesale-based option, basing it off of MSRP could create a gigantic loophole.
Cigar manufacturers could skirt the law by suggesting that every SKU carries an MSRP of $10 or more, but not adjust the wholesale price. Retailers could keep the current margins they currently have, while simply ignoring the suggested retail price.
In effect—nothing changes. One big loophole and one big reason why state’s don’t rely on the register price when it comes to regulation.
(For those wondering about sales tax, it’s less of an issue because of its utilitarian approach. In most states little cigars, premium cigars and cigarettes are taxed at very different tobacco tax rates, while they all have the same sales tax rate.)
3. THIS IS ALSO WHY “AFTER ANY DISCOUNTS” IS LIKELY GETTING DROPPED
The problem with the discounting standard is that it’s not anymore enforceable than the MSRP option.
Once the cigars are brought into the U.S. and shipped to retailers, they will be without the warning labels, compliance and other standards that would need to have been done before the cigars were sent to retail shelves. Discounting would mean that products without these labels or compliance checks could be sold for under $10 and it’s unclear how the FDA would legitimately control it.
Sure, you could create a sign at every retailer that says “In order for a premium cigar to be sold without a warning label it needs to be $10 or more,” but that seems unlikely and would also be contradicted by the grandfathered products.
The FDA certainly could punish retailers that violated this standard, but that would require random checks at every place where premium cigars are sold in the U.S. That’s thousands of random inspections, presumably required multiple times per year, indefinitely.
In other words—it’s not effective.
4. THE PUBLIC HEALTH MANDATE DOES NOT HELP
Suppose a manufacturer charges $10 for a cigar with a band, and $9.60 for one without the band. Both cigars contain the exact same types of tobacco and are rolled by the same person. The difference? The cigar band, a piece of paper used for decoration.
One product would be subject to the disclosure, compliance, warning labels, sample ban and others—while the other product would not.
The FDA’s point regarding premium vs. non-premium cigars is simple: it believes one product is not marketed to or purchased by minors. Given the organization’s public health mandate, it’s completely unreasonable to believe that it would be able to somehow suggest that two cigars, containing the same blend of tobacco, would be treated differently simply based off of price.
The way the FDA is explaining its justification for a premium cigar exemption will require it to show that the products that fall into the exempted category are actually different than those that don’t receive an exemption. If its definition of “premium cigar” fails to be defining and/or differentiating than the exemption will unravel, much like it does in the example above.
5. AND YES, $10 IS TOO HIGH
The FDA determined the $10 price point by determining what it believed was a representation of the market. Anyone that spends ample time in a humidor knows this probably isn’t the case.
Gary Griffith, better known for being the head of Emilio Cigar, manages retail humidors for Cigar-ette City and DOT Discount stores in four states. Based off of the 26 stores he manages, less than one quarter of sales would actually be exempt from the regulations.
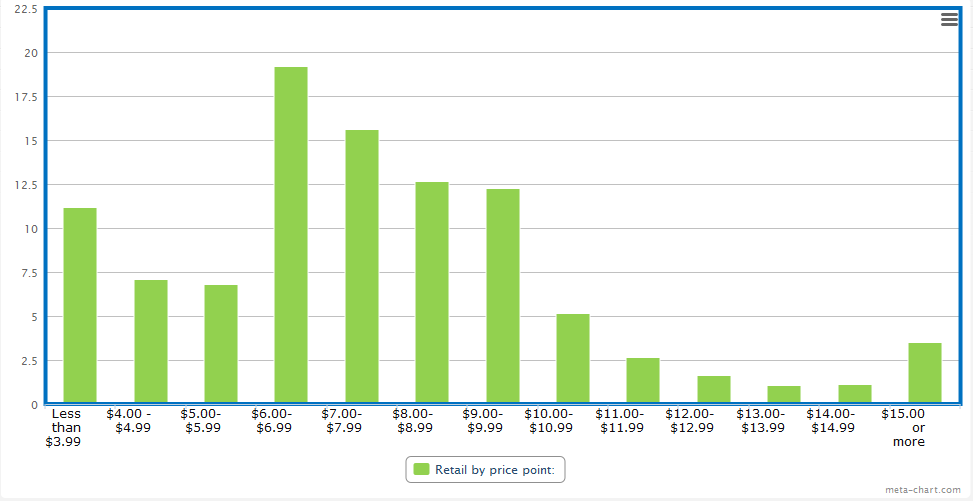
(The y-axis, the one on the left side, shows the percentage of sales)
Inevitably, there is no perfect price standard. The one the FDA settles on, if it goes that route, will either include some products the FDA intends on regulating, exclude products the FDA does not have interest in regulating, or in all likelihood some combination of both.
The state of New Hampshire defines “premium cigars” as:
Premium cigars are cigars made entirely by hand of all natural tobacco leaf, hand constructed and hand wrapped, wholesaling for $2 or more, weighing more than 3 pounds per 1000 cigars, and kept in a humidor at the proper humidity.
This definition would excluded products like Swisher Sweets or Black & Milds and include most products found in retail humidors, although some bundle cigars will not meet the requirement. If price is still a factor for defining “premium cigars” then some $2 bundle cigars are going to suffer at the expense of the rest of the humidor.
The attention solely on the $10 portion of requirement number six is misguided. For premium cigar smokers, it cannot simply be about “beating the FDA”—it has to be about establishing rules that not only the industry can live by, but also ones that will prevent the FDA from needing to revisit the category if an exemption is given.
Given the FDA’s concerns about little cigars, making sure that the definition of “a premium cigar” is written in a manner that clearly excludes them is crucial. That leads to what is the most challenging part of the current proposed definition, number seven.
7. Does not have a characterizing flavor other than tobacco; and
It should be very clear, flavored cigars are not banned, simply put—under the current definition, flavors cigars cannot be exempted as a premium cigar and as such will be subject to FDA regulation.
While the obvious reaction is “this is something that only affects Drew Estate and Miami Cigar & Co. (Tatiana),” the reality could not be further from the truth.
Rocky Patel (Java), J.C. Newman (Factory Throwouts), Davidoff (Baccarat), General Cigar (CAO Flavours), Nat Sherman (Host), Gurkha (Cojimar) and others all have brands on the U.S. market. Manufacturers, particularly those in the Dominican Republic, could be hurt by a reduction in sales from brands they produced. Thousands of retailers would be affected at their bottom line, as well of tens of thousands of adults, who could see their ability to enjoy these products go away.
This challenge will likely be more difficult. There will be more pressure and much more work needed to convinced the FDA that these types of flavored cigars are premium. That will likely see the humidification element come back into the discussion, as well as testimony from retailers and consumers explaining that an Acid is not the same as a Swisher Sweet.
The key to crafting a policy that ensures the future of the premium cigar industry is grounded in knowledge. It’s also one that should not be looked at like a war, particularly as the FDA as an enemy. At the moment, the organization is both judge and jury for premium cigars, yet, it’s also one that seems somewhat favorable at the moment to premium cigars.
Rather than framing the conversation with the regulatory body as “get out of our humidors; get off our lawn”—the premium cigar industry will be best served by explaining how the organization can exempt premium cigars without disrupting both the premium cigar industry, but also the FDA’s other goal.
The FDA always has the option for the last laugh. Option one.
For a more complete breakdown of the deeming document regarding premium cigars, click here.
To read the document in its entirety, click here. To submit public comment, click here.



